
Abstract:The properties of surface and internal materials after acid pickling of tantalum powder were com_ pared and analyzed, especially the changes of properties, chemical properties and electrical properties between tantalum powder surface and normal materials after acid pickling. The results show that the bulk density and Fischer particle size of the surface materials are larger than those of the normal materials, the metal impurity, content and leakage current of the surface material are obviously higher than those of the internal material.
Capacitor-grade tantalum powder is primarily used as the anode material in tantalum electrolytic capacitors. Because a dense, stable, and high-dielectric-strength amorphous oxide film can form on the surface of tantalum metal, and because applying different voltages in an acidic electrolyte allows for the formation of oxide films of controllable thickness, it is particularly suitable for the manufacture of small, high-capacity, and highly reliable capacitors. It is widely used in fields such as aerospace and telecommunications equipment [1–7]. To increase capacitance, tantalum powder must have smaller primary particles, a larger specific surface area, and a higher oxygen content. However, excessively high oxygen content—particularly oxygen accumulated on the surface of the tantalum metal during contact with oxygen-containing media—forms crystalline sites in the anodic oxide film, leading to increased leakage current and a reduced flashover voltage; On the other hand, finer-grained tantalum powder can result in narrower anode channels during capacitor manufacturing [8]. In particular, during vacuum heat treatment, surface oxygen diffuses into the interior of the tantalum powder, forming non-equilibrium dissolved oxygen. After surface activation, more oxygen is adsorbed to form tantalum oxide. High oxygen content during the sintering of tantalum anodes increases capacitor leakage current, reduces breakdown voltage, and causes the tantalum wire to become brittle. Therefore, the production process for tantalum powder used in capacitors requires further deoxygenation treatment of the powder [9].
It has been reported internationally that treating tantalum powder with oxygen-affinity metals such as calcium, magnesium, and aluminum can reduce its oxygen content. Typically, magnesium metal is used to reduce tantalum oxide, yielding tantalum powder and magnesium oxide as reaction products. Stark Company employs a method using gaseous magnesium to reduce tantalum oxide for the production of tantalum powder; experiments have demonstrated that the use of magnesium shavings reduces the tantalum oxide content in the powder. After oxygen reduction and removal from the furnace, surface material was collected from tantalum powder of different grades. Both the surface and internal materials were processed using the same acid washing procedure, and their physical and chemical properties were compared and analyzed, with a focus on differences in electrical properties. This provides a basis for effectively reducing leakage current in tantalum powder.
1. Experiment
1.1 Principle of the Deoxygenation Process
After vacuum heat treatment, the levels of metallic impurities (K) and gaseous impurities (H, C, etc.) in tantalum powder are reduced, and the particles become more compact. While the leakage current of the tantalum powder improves significantly, surface oxygen diffuses into the interior of the powder and remains trapped within the particles; a deoxygenation treatment is therefore required to achieve equilibrium. The deoxygenation process utilizes the principle of metal reduction of tantalum oxide to reduce the oxygen content in the tantalum powder. The heat-treated tantalum powder awaiting deoxidation is mixed with an excess of magnesium powder and loaded into a tantalum crucible for deoxidation. Under argon gas protection, the mixture is held at a temperature between 800 and 900°C for 1 to 4 hours to perform the deoxidation treatment. After the furnace temperature cools to room temperature, passivating gas is repeatedly introduced to slowly passivate the powder. After removal from the furnace, acid is added to remove residual metallic magnesium and magnesium oxide generated during the deoxidation process. The powder is then washed with pure water to remove various ionic impurities, dried, and sieved.
Chemical equation:
Ta2O5+5Mg→2Ta+5MgO
Mg+2H+→Mg2++H2
MgO+2H+→Mg2++H2O
1.2 Major Equipment and Raw Materials
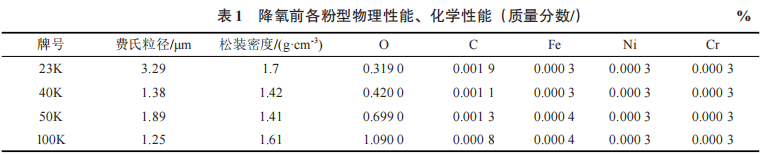
The main equipment used in the experiment includes pickling tanks, rinsing tanks, and vacuum drying ovens. The primary raw material is tantalum powder, including 23 K, 40 K, 50 K, and 100 K tantalum powders obtained after deoxidation and removal from the furnace, as well as surface material and internal material from the same batch (see Table 1). Nitric acid and hydrofluoric acid are of industrial purity.
Table 1 Physical and chemical properties of each powder type before deoxygenation (mass fraction/)
1.3 Test Protocol
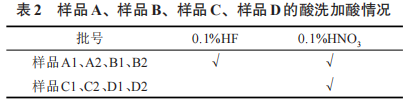
(1) After deoxidizing and removing from the furnace the four grades of tantalum powder—23 K tantalum powder (Type A), 40 K tantalum powder (Type B), 50 K tantalum powder (Type C), and 100 K tantalum powder (Type D)—2 kg of surface material was scraped from each. The surface material and the internal material were acid-washed according to the conditions in Table 2, and then sieved using the same drying and sieving procedures. After acid washing, drying, and sieving, the surface material samples were designated as A1, B1, C1, and D1, respectively, while the internal material samples were designated as A2, B2, C2, and D2, respectively.
Table 2 Acid addition during acid washing for Sample A, Sample B, Sample C, and Sample D
(2)Physical properties, chemical composition, and electrical properties of samples A1, A2, B1, B2, C1, C2, D1, and D2 were analyzed, and differences in their performance were compared. Because the deoxidation process involved the addition of excess magnesium, the impurity content present after deoxidation and removal from the furnace interfered with the results of physical and chemical testing; therefore, no sample analysis was performed on the surface or internal material after deoxidation and removal from the furnace.
1.4 Analysis and Testing
In accordance with the GB/T 1479-2011 and GB/T 3249-2022 standards, material performance testing was conducted using a Hall flowmeter, a WLP-202 average particle size analyzer, and a Scott cup, respectively. An LECO CS-436 oxygen-nitrogen analyzer manufactured by LECO CS (USA) was used to analyze O and N content in accordance with the GB/T 15076-2017 standard; the carbon-sulfur analyzer used was the CS-444 model manufactured by LECO CS (USA). Electrical property testing was conducted in accordance with the GB/T 3137-2020 standard using a ZRJ-30-22L bench-top vacuum sintering furnace, a 4339B High Resistance Meter for leakage current testing, and an E4980A Precision LCR Meter for capacitance testing.
2 Results and Discussion
2.1 Comparison of the Physical Properties of Surface and Internal Materials in Tantalum Powder of Different Grades
A comparison of the physical properties of samples A1, A2, B1, B2, C1, C2, D1, and D2 is shown in Figures 1 and 2. As can be seen from Figures 1 and 2, the surface material of the A, B, C, and D powder types has a larger Fritsch particle size and bulk density than the internal material. During the deoxidation process, as solid magnesium transforms into liquid and gaseous states, solid-liquid sintering and gas-solid reactions occur. When solid magnesium melts into a liquid state, the molten magnesium flows through the pores of the particles toward the bottom of the crucible. As part of the magnesium metal transforms into a gaseous state, the magnesium vapor travels through the pores of the tantalum powder and comes into contact with the upper crucible lid, condensing and flowing back onto the surface material of the tantalum powder. Here, the molten magnesium undergoes a liquid-solid sintering reaction with the surface material of the tantalum powder. The volatilized magnesium vapor encounters the thermal barrier at the top of the reaction vessel, condenses, and flows back into the vessel. Some of the refluxed liquid enters the gaps in the poorly sealed reaction lid, where it comes into contact with the surface material, thereby increasing the liquid-solid reaction contact area. The specific reaction mechanism is shown in Figure 3. Ultimately, this leads to stronger agglomeration of the surface material and the material at the bottom of the crucible, resulting in a larger Fritsch particle size for the product, reduced sintering porosity, and a higher bulk density of the product. Since the experiment only compared surface material with internal material, the product’s Fritsch particle size and bulk density were larger for the surface material than for the internal material.
Samples A1, A2, C1, and C2 were selected for a comparison of their scanning electron microscope (SEM) morphologies, as shown in Figure 4.
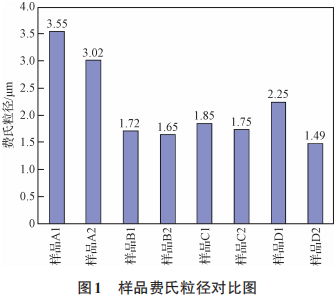
Figure 1 Comparison of sample particle sizes according to the Fritsch method
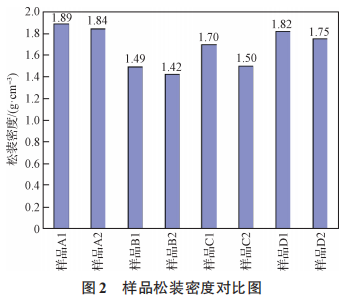
Figure 2 Comparison of Bulk Densities of Samples
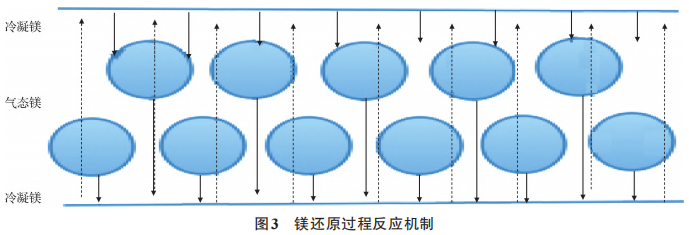
Figure 3 Reaction mechanism of the magnesium reduction process


Figure 4 Comparison of SEM morphologies of the sample at different magnifications
The SEM morphology of the surface material shows better sintering and agglomeration than that of the internal material, with a thicker sintering neck. The liquid magnesium metal on the surface interacts with the powder between particles, increasing the solubility of small particles in the liquid or solid phase. After the small particles dissolve, the solute precipitates at certain locations on the large particles, causing the large particles to grow larger and resulting in a thicker sintering neck. The current process leads to inconsistent sintering of tantalum powder in the liquid-solid phase pores during the deoxidation process. The existing deoxidation process needs to be improved to reduce the liquid-solid sintering reaction time and resolve the issues of agglomerate formation and inconsistent sintering diameters.
2.2 Comparison of the Chemical Properties of the Surface and Internal Materials in Different Types of Oxygen-Reducing Tantalum Powder
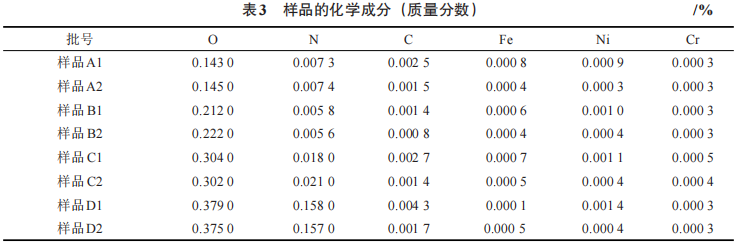
The physical properties of samples A1, A2, B1, B2, C1, C2, D1, and D2 are shown in Table 3. As can be seen from Table 3, the O and N contents of the surface and internal materials of the different types of oxygen-reduced tantalum powders are comparable, while the differences in metal impurities and C content are slightly greater. During the deoxidation process, the crucible lid did not seal tightly. Magnesium, being a chemically active alkali metal, interacted with the iron and nickel inside the reaction vessel at high temperatures, causing corrosion. This led to the surface tantalum powder adsorbing more iron and nickel corrosion products, resulting in significantly higher iron and nickel content in the surface material compared to the internal tantalum powder. The carbon content of the surface material is higher than that of the internal material. At temperatures of 800–1,100°C, organic carbon molecules come into contact with the surface of the tantalum powder where the oxide film has been disrupted and dissolve into the surface of the tantalum powder; therefore, the carbon content of the surface material is higher than that of the internal material. Hydrofluoric acid and nitric acid can effectively reduce the mass fraction of impurity elements such as carbon, iron, and nickel in tantalum powder [10], provided that the process is conducted in a high-concentration acid system, as high-concentration acid systems cause changes in the surface morphology of the particles. The use of low-concentration acid systems in the experiment fails to effectively remove impurities such as carbon, iron, and nickel generated during the deoxidation process. To prevent excessively high levels of iron, nickel, and carbon impurities in the final product caused by high impurity levels in the surface material, measures must be taken during the deoxidation process to address the issue of elevated carbon, iron, and nickel levels in the surface material of the tantalum powder.
Table 3 Chemical Composition of Samples (Mass Fraction)
2.3 Comparison of Electrical Properties Between the Surface and Internal Materials of Tantalum Powder with Different Oxygen-Reducing Formulations
Samples A1 and A2 were tested for electrical properties using FTA230 as specified in the GB/T 3137-2020 test method for electrical properties of tantalum powder; samples B1 and B2 were tested using FTA400; samples C1, C2 were tested for electrical properties according to FTA500 in the GB/T 3137-2020 test method for electrical properties of tantalum powder, and samples D1 and D2 were tested according to FTA100K in the GB/T 3137-2020 test method for electrical properties of tantalum powder. The test results are shown in Figures 5, 6, 7, and 8.
As shown in Figures 5, 6, 7, and 8, the surface material of the oxygen-reducing powders of types A, B, C, and D exhibits higher leakage current than the internal material, while other electrical parameters are comparable. During the growth of the anodic oxide film, the primary cause of film rupture is an abnormally high content of chemical impurities or the presence of inclusions, which results in poor film formation, leading to discontinuities in the oxide film or rupture at the impurity sites. A large number of electrons passing through the inclusions result in excessive leakage current or even breakdown. An increasing number of tantalum powder manufacturing experts and tantalum capacitor manufacturing experts believe that carbon in tantalum powder for capacitors is one of the most harmful impurities [11–13]. The presence of impurities in tantalum powder can cause the formation of second-phase particles in the base metal. During the anodizing process, these particles act as nucleation centers for the crystallization of the anodic oxide film, resulting in defects in the oxide film. As the carbon content increases, the leakage current of the tantalum powder tends to rise [14]. As the most harmful impurity in tantalum powder, carbon causes voids in the oxide film, leading to discontinuities in the film and resulting in leakage current. An increase in the content of any impurity will affect the performance of the capacitor, particularly by increasing leakage current [15]. The surface material of powder types A, B, C, and D has higher levels of metallic impurities and carbon content compared to the internal material, and therefore exhibits higher leakage current than the internal material.
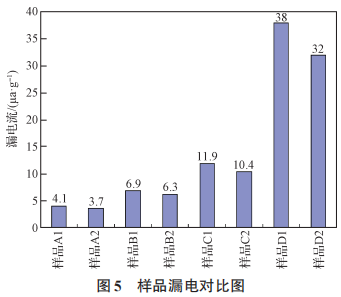
Figure 5 Comparison of leakage current in samples
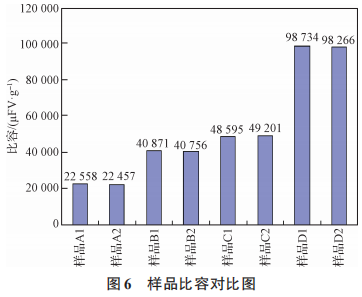
Figure 6 Comparison of Specific Volumes of Samples
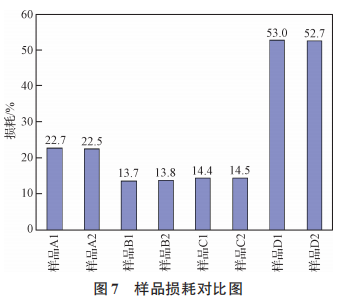
Figure 7 Comparison of Sample Loss
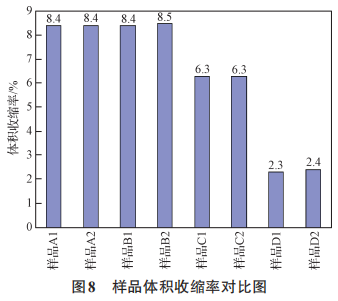
Figure 8 Comparison of Sample Volume Shrinkage Rates
3. Conclusions
(1) During the deoxidation process, the Frenkel particle size and bulk density of the surface material are larger than those of the internal material. The existing deoxidation process needs to be improved to reduce the liquid-solid reaction time and resolve the inconsistencies in agglomerate size and sintered particle size.
(2) The deoxidation process results in significantly higher levels of metallic impurities, carbon content, and leakage current in the surface material of tantalum powder compared to the internal material. To prevent these elevated levels of metallic impurities and carbon content in the surface material from adversely affecting the electrical performance of the final product, it is particularly important to control metallic impurities and carbon content during the deoxidation process.
References: Powder Metallurgy Industry, Vol. 34, No. 6, pp. 57–61, Dec. 2024; The influence of oxygen reducing tantalum powder surface fabric on the properties of tantalum pewder; LI Lijun ,LIN Fukun ,LI Zhenglong
With the rapid development of the high-end manufacturing industry, rare refractory metals and their powdered materials have become the cornerstone of sectors such as aerospace, electronics, and healthcare. Stardust Technology has established a strong presence in this field, specializing in the research, development, and production of high-end spherical powders made from rare refractory metals, while also providing end-to-end technical services. Leveraging its solid capabilities, the company is driving the industry forward. A comprehensive product range and reliable quality are among Stardust Technology’s core strengths. The company specializes in a variety of high-end rare refractory metals—including tungsten, molybdenum, tantalum, niobium, vanadium, and titanium—as well as their alloys and compound spherical powders, capable of meeting the diverse application needs of various industries. Leveraging advanced powder production technologies such as RF plasma spheronization, our products achieve industry-leading standards in purity, sphericity, and flowability. They meet the stringent material requirements of high-end processes like additive manufacturing, laying a solid foundation for downstream product quality. Our comprehensive technical service system facilitates deep integration between industry, academia, and research. Stardust Technology not only provides high-quality powder products but also offers one-stop technical services, including RF plasma spheronization, plasma rotating electrode atomization, additive manufacturing, and hot isostatic pressing, covering the entire process from material preparation to finished product processing. Relying on a professional R&D team and collaborative research institutions, the company overcomes key technical challenges in powder preparation and processing, providing customized solutions based on customer needs to enhance production efficiency and product competitiveness. For more product information, please contact our professional sales manager, Mr. Zhu, at 13378626726.
